About the Study
A prospective, multicenter, single-arm, open-label study to evaluate the safety and effectiveness of the Transdermal Compress bone-anchored implant in participants with transfemoral amputations.
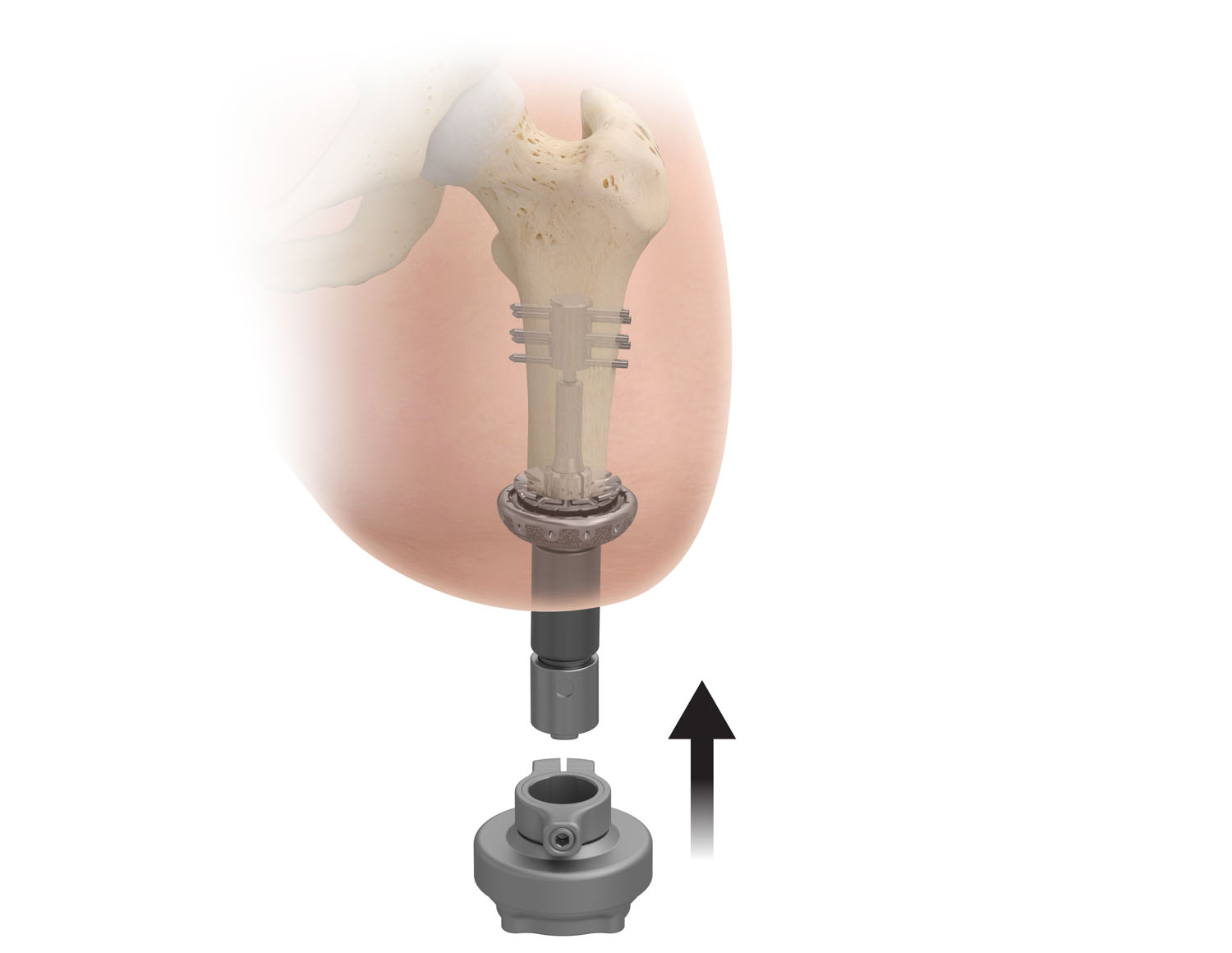

What is Transdermal Compress?
The Transdermal Compress investigational device is a bone anchored implant that allows for direct attachment of a prosthetic leg without the use of a socket. It is intended for above-the-knee amputees who experience problems with, or cannot use, a conventional socket prostheses.
Why Transdermal Compress?
- Single-stage surgery
- Use of established bone-anchoring technology
- May accommodate a wide range of limb lengths
- Prosthetic connector with resettable safety release available in multiple settings
About the Clinical Study
- Participants in this clinical study are committing to five years of study participation.
- Participants will be asked to complete questionnaires regarding prosthetic use and overall health using a study app on their phone.
- The Transdermal Compress investigational implant and the investigational Resettable Torque Connector will be provided at no cost to study participants.
- Please discuss full participation requirements and study risks with your care team.
Additional Considerations
The study rehabilitation protocol must be followed closely and will be directed by your surgeon, a physical therapist, and prosthetist.
The implant does not have the same characteristics as normal, healthy bone. Participants will be asked to avoid high impact activities.
